Molecular Solution
Water
Water molecular models are computational techniques that have been developed in order to help discover the structure of water. In this section, you will be asked to calculate some physical properties based on classical molecular dynamics simulation. The TIP3/Fw model will be usded in the simulations.
We have prepared a CP2K input file water.inp for running a MD simulation of liquid water using the force field from the first exercise (parametrized by Praprotnik et al.).
- Check that the MD is energy conserving and well-behaved.
- What are the final average temperatures in each of the simulations? Why are they different from the initial ones?
- The initial atomic configuration stems from an equilibration run. At which temperature was the system (approximately) equilibrated?
Next we are going to analyze the trajectories in order to calculate the radial distribution function (rdf, $g(r)$) as a function of distance $r$.
VMD comes with an extension for exactly this purpose: In the VMD Main window open “Extensions → Analysis” click on “Radial Pair Distribution function $g(r)$”. In the appearing window use “Utilities → Set unit cell dimensions” to let VMD know the simulation box you used. After that use Selection 1 and 2 to define the atomic types that you want to calculate the rdf for, for example “element H”.
- Plot $g_{O-O}(r)$ at 300K and experimental value
goo.ALStaken at 300 K into same graph.
Then we will calculate diffusion coefficient. It is a proportionality constant between the molar flux due to molecular diffusion and the gradient in the concentration of the species (or the driving force for diffusion), which is defined by: $6D=\lim_{t\to\infty} \ \frac{\delta <r^2(t)>}{\delta t}$
To evaluate this expression, all that is needed is to evaluate at each point in time in the calculation the average of the square of the distance that each atom has traveled since the start of the production phase of the dynamics, and examining the slope of this function at a long time. By storing the initial coordinates, it is straightforward to evaluate the square of the distance. However, some care is needed due to the use of periodic boundary conditions: the program stores x, the coordinates, but in many programs, during the dynamics, if any atom has its x, y, or z coordinate become larger than box size or smaller than zero, then it is moved back into the other side of the box. This has the effect of making the raw distance traveled meaningless. The value of D is obtained from the slope, at a long time, of the right-hand side of the above equation (you need to divide by six to obtain D, take the slope, and also be careful with units).
VMD comes with an extension for exactly this purpose: In the VMD Main window open “Extensions → Analysis” click on “RMSD Trajectory Tool”. In the appearing window use “all” to let VMD know the molecule you want to track. Tick “Plot”, and press “RMSD”, you will have the RMSD plot for the water system.
- Plot RMSD for the water at 300K and calculate corresponding diffusion coefficient, are they expected?
We will compute the vibrational spectrum, and dielectric constant of water based on molecular dynamics. The spectra for water are available in this paper https://doi.org/10.1063/1.1884609. The provided program computes the correlation function of the (derivative of) the dipole moment and performs the Fourier transform.
\begin{equation} A(\omega)\propto{\int\langle{\mu}({\tau}){\mu}(t+{\tau})\rangle_{\tau}e^{-i{\omega}t}d{t}}, \label{eq:auto} \end{equation}
The dielectric constant of a system describes its response to an external electric field.
If the dipole moment is properly sampled, one can compute the dielectric constant of water, by applying the Kubo Formula. This is valid in the approximation that the response of the system to the time-dependent perturbation (the field) is linear.
The dielectric constant can then be calculated from the dipole moments via: \begin{equation} \epsilon = 1 + \left(\frac{4 \pi}{3 \epsilon_0 V k_B T } \right ) \operatorname{Var}(M) \ , \end{equation}
where $M$ denotes the dipole moment of the entire simulation cell and $\operatorname{Var}(M)$ denotes the variance of the dipole moment of the sampling: \begin{equation} \operatorname{Var}(M) = (\langle M \cdot M\rangle - \langle M\rangle\langle M\rangle ) \ . \end{equation}
Compile the FORTRAN code, and execute the program
gfortran cpt_ir_diele.f90 -o cpt_ir_diele.o ./cpt_ir_diele.o < dipole.in
- Compute the IR spectrum and plot it, match the frequencies with vibrational mode.
- Compute the dielectric constant of water at 300K.
- Does IR or dielectric constant match the experimenal value? If not, why?
Ramachandran plot
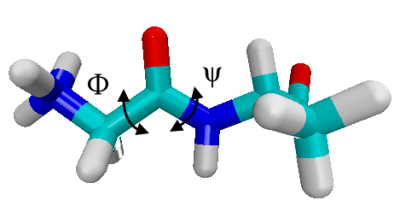
Glyala is one of the simplest molecules that exhibits some important features common to larger biomolecules. In particular, it has more than one long-lived conformation, which we will identify in this exercise by mapping out its potential energy surface.
The conformations of glyala dipeptide are characterized by the dihedral angles of the backbone. Below, we color carbons in green, hydrogens in white, oxygen in red and nitrogen in blue, i.e. the torsional angle $\phi$ is N-C-C-N , while $\psi$ is C-N-C-C along the backbone.
tar -xvf glyala-epot.tar.gz
glyala.pdb with VMD and determine the atomic indices of the atoms defining the dihedral angles.
With this knowledge at hand, we will fix the dihedral angles and perform geometry optimization for all remaining degrees of freedom.
- The atomic indices defining the dihedral indices in the input file
geo.inare missing. ReplaceI1toI4by the atomic indices determined previously. - Use
perform-gopt.shto perform the grid of geometry optimizations. - Use gnuplot to plot the potential energy surface (we have provided a script
epot.gp). Which are the two most favoured conformations?$ gnuplot
gnuplot > load "epot.gp"
Glyala in water
Now, we will move to a more realistic system - Glyala in water. We will preformed a MD of glyala in water and save the trajectory.
The initial geometry provided in the PDB file is a glyala molecule solvated by 73 water molecules. The geometry is not equilibrated. You need first to equilibrate the system at 300K. When the system is equilibrated, you need to analysis the result.
- Perform the molecular dynamics simulation using NVT ensemble at 300K. Change TIMECON (i.e.500, 2000 fs) in the &THERMOSTAT section.
- Determine from which step the system is equilibrated, plot the calculated properties and explain why.
- Compute the O-O radial distribution function for water with acceptable statistics using 20 ps (after equilibration) of simulated time.
- Determine the solvation shell by calculating RDF of g$_{CO}$ (carbon atoms from glyala and oxygen atoms from water)
In last exercise, one already knew how to calculate the RDF for the Argon system. In TASK3, you need to calculate the RDF only for water instead of whole system. Since the glyala contain two oxygen atoms, it is not reasonable to include the oxygen atoms in glyala molecule if we are only interested in O-O RDF for water. Using VMD, the O-O RDF for the water can be easily calculated. In the
Selection 1, Selection 2
, one need to specify
element O and not same residue as element C
The frames should start from the beginning of production run.